Kennedy Rare Earth Element Project
Location
The 100%-owned Kennedy Rare Earth Element project encompasses two granted EPMs and 10 EPM applications in North Queensland.
North Queensland hosts a variety of intrusive and metamorphic rocks that contain elevated rare earth elements. When these rocks are weathered, especially under hot, humid conditions, they contribute to proximal, residual clay or fine sediment deposits. These may in turn be subject to further weathering and formation of laterite. During these weathering process rare earth elements may adhere to and concentrate in clays.
The key to viability of this style of deposit is low cost of beneficiation. Rare earth elements can be extracted by simple leach, desorption chemistry using weak acids in ambient conditions.
Initial metallurgical work on early drill samples has already indicated favourable results both with respect to rare earth element recoveries and acid consumption.
Exploration
Previous preliminary metallurgical test work from the project indicates that a significant proportion of the near-surface rare earth mineralisation has formed as Ionic Adsorption REE Clays, with leach testwork demonstrating that rapid recoveries can be achieved by desorption of REE’s in the first 30 minutes using a weak acid (pH4) ammonium sulfate solution ((NH4)2SO4).
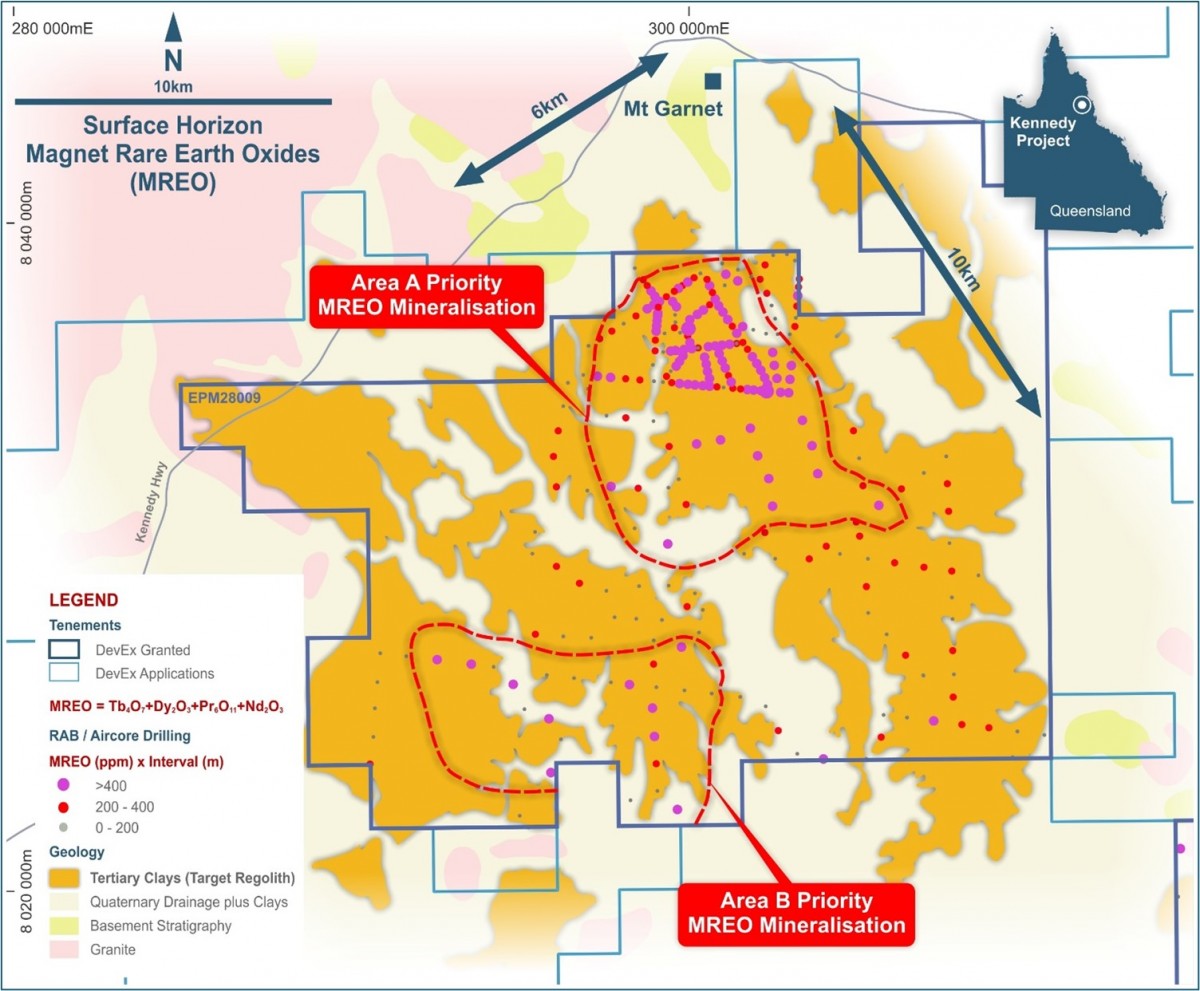
Figure 1. Shallow air-core drilling and MREO grades (ppm) x Interval (m) from the Surface Horizon within the Target Regolith. Two priority MREO areas comprising higher MREO-grade and widths are apparent in the drill data.
Assay results from 2023 air-core drilling has expanded the footprint of total rare earth oxide (TREO) mineralisation in surface clays, with higher grade intervals including:
- 3m @ 1,370ppm TREO (incl 219 ppm MREO) from surface (KAC099)
- 4m @ 1,056ppm TREO (incl 181 ppm MREO) from surface (KAC114)
- 3m @ 1,190ppm TREO (incl 210 ppm MREO) from surface (KAC116)
- 2.5m @ 1,155ppm TREO (incl 197 ppm MREO) from surface (KAC216)
- 3.5m @ 1,208ppm TREO (incl 196 ppm MREO) from surface (KAC232)
- 3.5m @ 1,237ppm TREO (incl 214 ppm MREO) from surface (KAC238)
- 3.5m @ 1,071ppm TREO (incl 209 ppm MREO) from surface (KAC243)
- 3m @ 1,039ppm TREO (incl 192 ppm MREO) from surface (KAC262)
- 3m @ 1,047ppm TREO (incl 194 ppm MREO) from surface (KAC312)
Importantly, shallow TREO assay results include the important and high-value MREO’s – Praseodymium (Pr6O11), Neodymium (Nd2O3), Dysprosium (Dy2O3) and Terbium (Tb4O7), which are essential in the manufacture of permanent rare earth magnets used in electric vehicles and numerous other renewable energy applications.
Next Steps
The Company has identified two priority areas where drilling has defined higher-grade MREO mineralisation in surface clays, with the northern area (Area A) extending over an area of up to 10km x 6km. The two priority MREO areas lie within a much broader region of TREO mineralized clays that remain only partially tested on broad drill spacing.
Drilling within both priority areas has identified that the tertiary clays (Target Regolith) which host the target MREO’s are typically unconsolidated loose clays mixed with poorly sorted pisolite material.
As part of the Queensland Government’s Collaborative Exploration Initiative to contribute to the discovery of critical minerals, DevEx was recently awarded $175,000 (excluding GST) to carry out further metallurgical sampling at the Kennedy Project.
For the latest project overview, view our ASX Announcements and Investor Presentation.
